This week’s Cover Story represents what we expect to be an ongoing collaboration for the benefit of the community between Connection Media, LLC, the parent company of Weld for Birmingham, and the Alabama Initiative for Independent Journalism, the newly formed nonprofit behind BirminghamWatch.org. The goal of this partnership is to provide stronger, deeper, more meaningful journalism in the public interest, carried out with professionalism and careful attention to ethics and journalistic integrity.
Our first project together concerns the ways society and the law are changing around the controversial topic of marijuana. “America’s Weed Rush,” a project originating with News21, the cornerstone of the Carnegie-Knight Initiative on the Future of Journalism Education, has been produced locally by BirminghamWatch and Weld. This week’s Weld includes stories from our local collaboration.
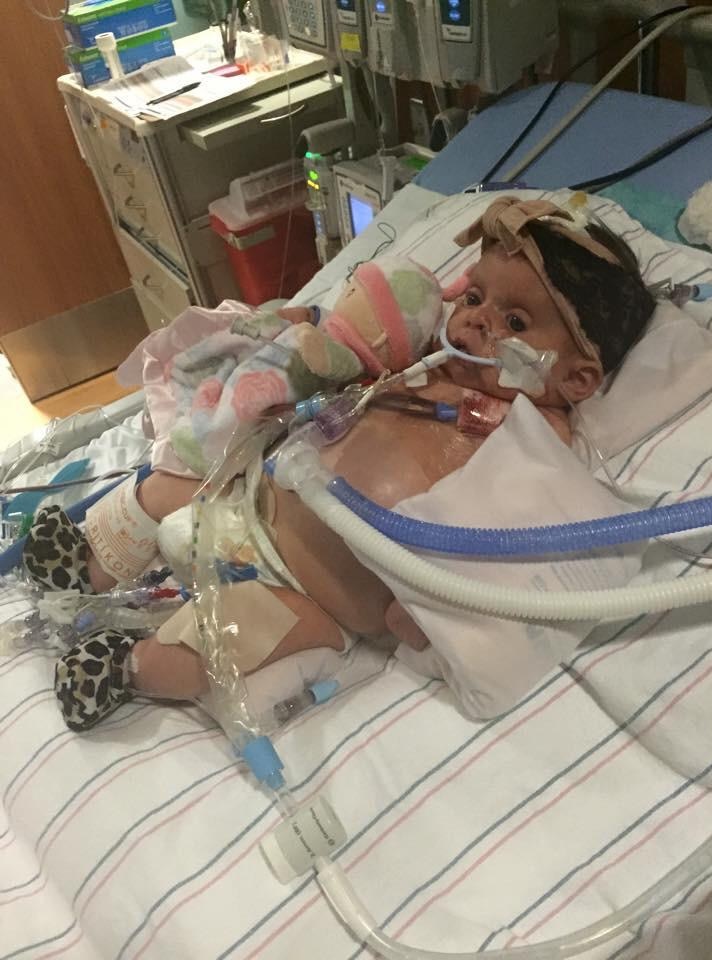
More than 50 patients, including adults and children, are now enrolled in University of Alabama at Birmingham studies to test the safety of a marijuana derivative that has shown promise as a treatment for severe epileptic seizures, according to university officials.
The two studies at UAB make up one of the largest research efforts in the nation into the medical use of cannabidiol, or CBD oil. The research promises to add to the assessment of a treatment now supported by anecdotal evidence but not yet shown scientifically to be a successful treatment.
Trial studies of CBD oil also are taking place at the University of Colorado, University of Iowa, University of Kentucky and University of Louisville.
The UAB School of Medicine began earlier this year to assess the safety of the supplement derived from the cannabis plant. CBD oil is given orally to patients in the study.
The research, which includes an adult study at the University of Alabama at Birmingham and a pediatric study at Children’s of Alabama, was authorized by the Alabama legislature in 2014 through legislation known as Carly’s Law. In Alabama and across the country, parents pushed for access to the treatment for their sick children.
So far, 51 patients — 27 adult and 24 pediatric — are participating in the research, UAB officials say.
Enrollment began in March and plans are to register 100 subjects, 50 adult and 50 pediatric. The studies test the safety of CBD oil in patients with intractable seizures, according to university officials.
“The two studies are gathering data on the overall patient experience, treatment outcomes and adverse effects,” said David Standaert, chair of the UAB Department of Neurology. “This is research that UAB is uniquely qualified to conduct and assess as we search for therapies for our patients with debilitating epilepsy.”
The research is a U.S. Food and Drug Administration–designated “expanded access” program, which “allows patients with intractable seizures access to this potentially beneficial therapy during the investigational stage,” Standaert explained.
UAB’s research monitors patients using the CBD for side effects as well as interactions with other medications. Patients take the CBD oil twice a day and are monitored by UAB officials for signs of progress. Patients come in for visits every two weeks, at which time an increased dosage can be prescribed.
The primary goal is to assess the safety and tolerance of the drug, but researchers also will be watching to see if patients with epilepsy who haven’t responded to conventional treatment experience any decrease in the number of seizures.
Bob Shepherd, a media specialist at UAB, said it’s far too soon to draw any conclusions from the ongoing studies. Most of the existing information about cannabidiol as a treatment for epilepsy is anecdotal. The UAB study could help determine the drug’s effectiveness for patients who don’t respond to other treatments.
“What we learn from these investigations could have a profound impact on the lives of many adults and children with uncontrolled seizures,” Standaert said.
School officials stress that their study of a marijuana derivative is not the same as research into “medical marijuana.” The FDA says the term medical marijuana refers to the whole unprocessed marijuana plant or its crude extracts, “which are not recognized or approved as medicine” by the FDA.
Still, scientific study of marijuana’s active chemicals, called cannabinoids, “… is leading to the development of new pharmaceuticals that harness the therapeutic benefits of cannabinoids while minimizing or eliminating the harmful side effects,” including the “high” produced by eating or smoking marijuana leaves, according to the FDA website.
About 40,000 people in Alabama suffer from epilepsy. The state’s first two patients in the legislation-backed Carly’s Law study received the medicine in spring 2015. A pediatric patient received the first dosage of CBD on April 1, and the first adult patient started the treatment a week later.
Carly’s Law is named for the daughter of Hoover’s Dustin and Amy Chandler, who pushed to allow the study that could potentially benefit children and adults, including their daughter, who has a rare genetic disorder called CDKL5. The program at UAB is structured in the law as a study to examine the drug’s effectiveness.
Carly is now part of the study. Her father said she has seen “great improvement” from the treatment.
To be considered for participation, potential study subjects must apply to the UAB Cannabidiol Program and meet specific qualifications in order to be enrolled. Information on how to apply for enrollment can be found on the UAB Cannabidiol website: uab.edu/cbd.
For related stories, visit, birminghamwatch.org